Cronobacter - Assurance GDS®
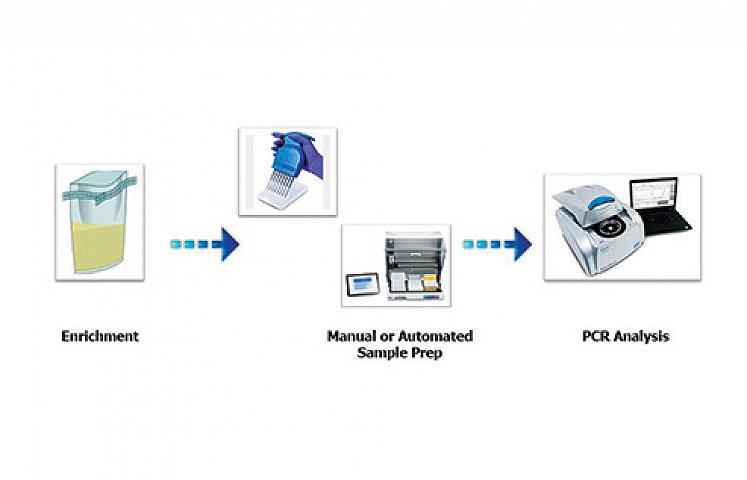
Assurance GDS Cronobacter Tq II assay is used for the detection of Cronobacter spp. in infant formula, ingredients, and environmental samples. Results are available in as little as 22 hours and holds both MicroVal and AOAC approvals.
The harmonised enrichment procedure between GDS Salmonella and Cronobacter Tq II will help laboratories save time and costs. The enrichment time also allows for increased overlap times between assays, which aid workflow flexibility and reduces the potential for human error between protocols.
Like other Assurance GDS Assays, the Cronobacter assay uses the PickPen Immunogenetic separation technology for pathogen enrichment. Magnetic particles coated with antibodies specific to the target pathogen capture and bind these. By separating the pathogens from the inhibitory background and concentrating them, the sensitivity of the assay is significantly improved. After washing the magnetic particles are released and transferred to the amplification tubes for PCR analysis, with each tube containing all the reagents in a ready to use pellet.
Assurance GDS for Cronobacter Features:
- Results available 22-26 hours
- Combined enrichment for Cronobacter and Salmonella saving time and reducing labour costs
- Simple 1 step enrichment procedure
- Use of PickPen to improve accuracy and prevent inhibition
- Accuracy of Assurance GDS proves comparable to ISO 22964:2017 reference method for the detection of Cronobacter spp. in foods and environmental samples
Details:
- Detects Cronobacter spp.
- AOAC Performance Tested Method Certificate Number: 121903
- MicroVal Certificate Number: 2017LR77
- Technology: Polymerase Chain Reaction
- Storage: 2 - 8⁰C
Merck Assurance GDS Key Advantages Flyer
Assurance GDS Cronobacter Tq (Qty 100 tests)
Code: 71038-100